Discovery of a tick ferritin 2, a novel iron-transporting protein as a candidate for anti-tick vaccine
Hajdušek O., Sojka D., Kopáček P., Burešová V., Franta Z., Loosová G., Grubhoffer L.
Research on how the hard tick (Ixodes ricinus), the vector of tick-borne encephalitis and Lyme boreliosis, manages the surplus of iron originating from the host blood led to the discovery of yet unknown protein designated as ferritin 2. In contrast to the previously described ferritin 1 functioning as intracellular iron-storage molecule, the novel ferritin 2 is secreted into the body-fluid (hemolymph) of the tick. The roles of ferritin 1, ferritin 2 and ‘iron-regulatory protein’ (IRP) in tick iron metabolism was studied using a method of RNA interference, which makes possible to specifically block their synthesis. Surprisingly, it was demonstrated for the new ferritin 2, that its main role is in iron transport from the tick gut to the peripheral organs like salivary glands or ovaries. Impairment of iron metabolism had a negative impact on tick reproduction and development. The most important result of ferritin 2 elimination was the suppression of tick feeding ability, by which half of the ticks died during blood feeding on the host. A similar effect was achieved by experimental vaccination of rabbits with recombinant ferritin 2, where the antibodies imbibed with the host blood efficiently blocked ferritin 2 in the tick gut. This fact, together with a remarkable molecular difference from ferritins of mammalian hosts, make the tick ferritin 2 a promising candidate for an efficient ‘anti-tick’ vaccine protecting from tick feeding and limiting their ability to transmit pathogens causing infectious diseases. The use of ferritin 2 as potential veterinary vaccine is protected by a Czech and an international patent claim. The pilot tests of this vaccine on cattle were performed in co-operation with a partner laboratory abroad and the results achieved supported further development towards commercialization of the vaccine and its broad application especially in countries, where the economical losses caused by ticks in livestock production are enormous.
Hajdušek O., Sojka D., Kopáček P., Burešová V., Franta Z., Šauman I., Winzerling J., Grubhoffer L.:
Knockdown of proteins involved in iron metabolism limits tick reproduction and development.
Proc. Nat. Acad. of Sci. U.S.A. 106, 1033-1038 (2009)
Hajdušek O., Almazan C., Loosová G., Villar M., Canale M., Grubhoffer L., Kopáček P., de la Fuente J.:
Characterization of ferritin 2 for the control of tick infestation.
Vaccine 28, 2993-2998 (2009)
Czech patent claim: Kopáček P., & Hajdušek O.: "Feritin 2 pro imunizaci organismu proti klíšťatům." Úřad průmyslového vlastnictví České republiky, PV 2008-402, (25.6.2008)
International patent claim treaty: Kopáček P., & Hajdušek O.: "Ferritin 2 for the host immunization against ticks." Úřad průmyslového vlastnictví České republiky, PCT/CZ2009/000085, (18.6.2009)
Panel A: Effect of RNA interference on the tick ability to feed on the host
gfp – ticks injected with control (GFP) double stranded RNA
fer1 KD – effect of intracellular ferritin 1 silencing
irp KD – effect of iron-regulatory protein (IRP) silencing
fer2 KD – effect of secreted ferritin 2 silencing
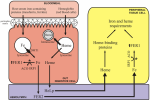
Panel B: A model of iron metabolism in ticks
Based on our results and work of Pedro L. Oliveira‘s laboratory (Rio de Janeiro) we assume two independent pathways for metabolism of heme (the blood pigment) and non-heme iron from the host blood. Silencing of ferritin 2 impairs transport of the non-heme iron, which is necessary e.g. for the respiratory enzymes. Moreover, the toxic iron is accumulated in the tick gut, which has a severe impact on tick ability to feed and develop.